Section
1: What is Health Food in China?
Definition of Health Food in China and in the World
The definitions of health food are different all around the world. In general, food products in below names are regarded as health food in China:
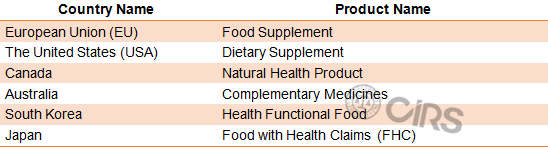
According to Food Safety Law of the People's Republic of China (2015 version), health food refers to food products which claim the health function based on scientific basis, and have no acute, sub-acute or chronic hazards to human body.
Categories of Health Food in China
In Chinese industry, health food is usually divided into two categories as following:
I. Nutrition supplement:
Health food that provides vitamins and/or minerals but without providing energy or other active ingredients.
II. Functional health food:
Health food that is claimed with health function and has physiological effects on the human body.
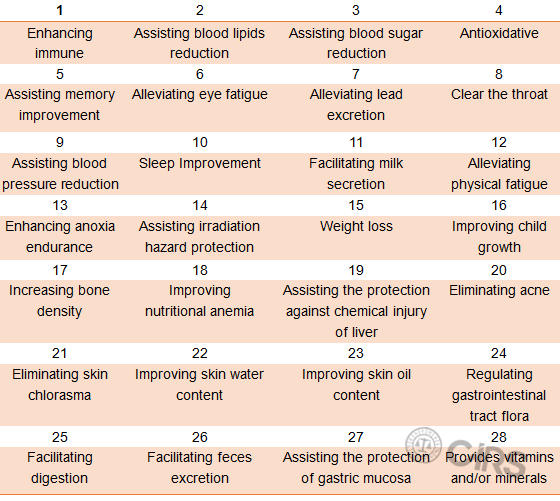
Approved Health Functions for Health Food in China
In accordance with current
Chinese health food regulations, there are 27 health functions for functional health
food and 1 health function for nutrition supplement.
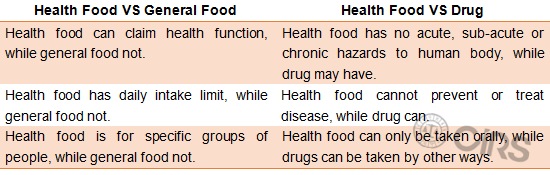
Differences among Health Food, General Food and Drug
Initially, health food was regarded as traditional Chinese medicine under drug category. Afterwards, people found that its properties were closer to properties of food rather than properties of drug. Therefore, it has been categorized under food category. However, it is definitely different from general food or drug.
Section 2: How Many Health Food Registered in China?
Since the implementation of the health food registration policy in 1996, there has been 17,279 approved health foods by the end of 2017, including 16494 approved domestic health foods and 785 approved imported health foods. During the recent 30 years, the approved health food amounts differently at different stages because of the changing policies. The numbers of approved health food with different function claims are different as requirements for 28 health function claims are different.

CIRS is making statistics of the approved health foods from different angles such as the year, health function, applicant, etc.
Section 3: Chinese Health Food Regulation in Present and Future
In the past two years, Chinese health food regulation has changed a lot. Former China Food and Drug Administration (CFDA) finally set up a complete regulatory system and had the appropriate policy and regulatory frameworks in place for health food in 2017. The new policy opened an access of registration and filing double-track system for health food.

Based on the public speaking from former CFDA leaders and communication with relevant officials, CIRS is forecasting the future policy as well. The detailed health food regulations and the trend of future policy will be presented by CIRS.
Section 4: Timeline and Budget for Health Food Access to the Chinese Market
According to the latest Chinese health food regulation, oversea companies who plan to place health food on Chinese market must apply for and obtain a Health food approval certificate before exportation to China. And all imported health food shall carry out the registration and filing certificate application with former CFDA. Benefit from the implementation of the new policy-filing, imported vitamin and mineral nutrition supplements application cost less time and money than functional health food.
The top concerned themes including required dossiers, process, timeline and budget for each step of health food registration and filing will be comprehensively collated by CIRS.
Section 5: How does Chinese Government Supervise the Health Food?
Different from the rules for admittance into Chinese health food market, the supervision of health food is becoming stricter in China. Former CFDA will do casual inspections more frequently, and will release all results to the public. In a word, the competent authority has less tolerance on illegal health food.
CIRS is counting illegal cases from 2015 to 2017. Combination with the relevant regulations, some typical food safety incidents will be pointed out.
Section 6: Cross-Border E-Commerce (CBEC): New Opportunity for Imported Health Food to Enter the Chinese Market
Due to the promotion of e-commerce policies, the rapid development of various e-commerce platforms, and the improvement of logistics networks, Cross-Border E-Commerce (CBEC) has become an important channel for the sale of imported health food, particularly for new brands.
In the recent years, the regulations and policies pertaining to CBEC have been changed for several times, CIRS is collecting the regulations and CBEC exportation requirements in details.
The summary of each section in the Guideline is introduced by a series of articles which are available as following. If it is of your interest, please kindly click to get information.
Coming soon
How Many Health Food are Registered in China?
Chinese Health Food Regulation in Present and Future
Timeline and Budget for Health Food Access to Chinese Market
How does Chinese Government Supervise the Health Food?
Cross-Border E-Commerce (CBEC): New Opportunity for Imported Health Food to Enter the Chinese Market
In addition, if you would like to get the
Editor:
CIRS Food Technical Team: Established in 2012, CIRS Food Technical Team has more than 80% masters with degrees of Food Safety or Food Engineering and more than 50% members from oversea renowned universities. Since it was founded, based on the rich experience of regulatory compliance and deep understanding of Chinese food regulations and industry, food specialists have made comprehensive and reasonable solutions for many oversea food companies to complete Chinese food regulatory compliance progress.
If you have any other questions, please contact us at service@jianzaoshiwang.cn.
Related Article
Cross-Border Acquisition: Chinese-funded Enterprises Frequently Acquire Foreign Health Food Brand
Data Analysis: Why are Health Food Enterprises Keen to Explore E-Commerce Channel?

